Improved classification of rare lymphoid neoplasms would be aided by a deeper understanding of their underlying molecular features and is important for diagnosis, prognosis and therapy. Tumor entities classified within the WHO category of splenic B cell lymphomas and leukemias often exhibit heterogenous, transecting features, and include hairy cell leukemia (HCL), splenic diffuse red pulp lymphoma (SDRPL), splenic marginal zone lymphoma (SMZL), and the newly described WHO entity, splenic B cell lymphoma/leukemia with prominent nucleoli (SBLPN); the latter including patients formerly classified as HCL-variant (HCL-V). Genome-wide epigenetic information provides a tumor cell fingerprint combining cell-of-origin and tumor-specific events. Here we used DNA methylation to perform an unbiased molecular subclassification and to explore novel biological aspects of these patients.
Samples from patients with a pathological diagnosis of HCL, HCL-V, SDRPL and SMZL (made prior to the 5 th WHO revision and ICC classifications) were obtained from 19 institutions across 9 countries, totaling 367 patients. Cells were FACS-purified where necessary and DNA was analyzed by 450/850K Illumina DNA methylation arrays. Genetic mutations were assessed by whole-exome or targeted sequencing, IGHV-D-J sequences by Sanger sequencing, and copy number alterations (CNAs) by Illumina arrays. The 1000 most variable CpG methylation sites were used for k-means clustering. Recursive feature elimination/random forest algorithms were used to develop a classifier for DNA methylation-based subgroups with 98% accuracy.
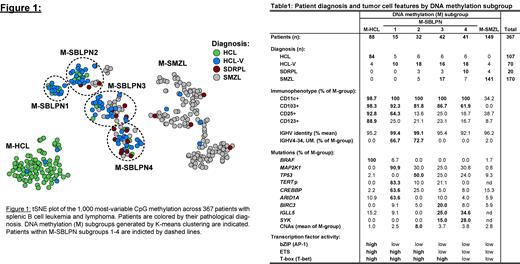
Unsupervised clustering of 197 patients diagnosed with HCL, HCL-V or SDRPL revealed 5 distinct DNA methylation (M) subgroups ( Figure 1). Subgroup assignment was stable throughout longitudinal sampling (including pre/post-treatment) and consistent between splenic, bone marrow and PBMC derived cells. A subgroup with universally clonal BRAF-V600E mutations and majority diagnosed as HCL was termed the M-HCL subgroup ( Table 1). Four other groups termed M-SBLPN1-4 contained all HCL-V and SDRPL diagnosed samples and were devoid of BRAF-V600E mutations. M-SBLPN1 comprised MAP2K1 mutations (91%) and was enriched for CREBBP, ARIDIA and TERT-promoter mutations. These patients displayed an HCL-like immunophenotype (64.3% CD25+) with 1/3 diagnosed as HCL. M-SBLPN2 exhibited the highest prevalence of TP53 mutations and concomitant genomic instability. Patients in M-SBLPN1,2 were enriched in unmutated IGHV4-34 rearrangements. M-SBLPN3,4 subgroups displayed an immunophenotype more dissimilar to HCL, mutated IGHV genes, and enrichment of IGLL5, SYK and BIRC3 mutations. M-SBLPN4 contained the most SDRPL samples, suggesting it may represent the SDRPL entity retained by the WHO. We next uncovered that 29/170 SMZL patients displayed DNA methylation patterns mapping to M-SBLPN2-4. These patients were phenotypically and molecularly similar to SBLPN (70% displaying villous morphology and depleted in IGHV1-2*04, NOTCH2, KLF2 mutations), likely representing SMZL patients suggested for reassignment to SBLPN in the updated WHO classification.
To elucidate molecular pathways governing the biology of M-SBLPN subgroups, transcription factor motif enrichment analysis in hypomethylated genomic regions revealed selective activation of AP-1 in M-HCL along with ETS in M-SBLPN1,2. Both transcription factors are downstream of MAPK signaling, consistent with activating BRAF and MAP2K1 mutations in these subgroups. However, we observed strong ETS enrichment in the absence of MAP2K1 in M-SBLPN along with mutual exclusivity of MAP2K1 and TP53 mutations, suggesting TP53 mutations are driving ETS activation. Although lymphoid neoplasms rarely exhibit TERT promoter mutations, 83% of M-SBLPN1 patients showed the c.-124C>T mutation commonly observed in other cancers producing an ETS binding site and ectopic TERT activation. ETS activation and gain of an ETS site by mutation implies oncogenesis involves aberrant TERT activation in this subgroup.
In summary, we have developed a DNA methylation-based classifier that resolves 4 SBLPN subgroups with distinct molecular features, and reclassifies a subset of SMZL and HCL patients, adding further information to the updated WHO/ICC entities. We reveal distinct biological pathways operating in M-SBLPN subgroups that may aid targeted therapy approaches.
Disclosures
Abdelbaky:Roche: Ended employment in the past 24 months. Baer:MLL Munich Leukemia Laboratory: Current Employment. Meggendorfer:MLL Munich Leukemia Laboratory: Current Employment. Bhat:AstraZeneca: Consultancy, Research Funding; Abbvie: Consultancy; Aptitude Health: Honoraria. Rogers:Pharmacyclics: Consultancy; AstraZeneca: Consultancy; Janssen: Consultancy; Novartis: Research Funding; Loxo@Lilly: Consultancy; Genentech: Consultancy, Research Funding; Beigene: Consultancy; AbbVie: Consultancy, Research Funding. Zenz:Beigene: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Lilly: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; AstraZeneca: Consultancy, Honoraria; Roche: Consultancy, Honoraria. Blachly:Leukemia Diagnostic Device: Patents & Royalties: Being prosecuted; AbbVie: Consultancy; Epigenetic classification of leukemia: Patents & Royalties: PCT conversion filed; AstraZeneca: Consultancy; Astellas: Consultancy. Zent:AstraZeneca: Research Funding; GenMab: Research Funding. Banerji:Biogen, U of M: Patents & Royalties: GSK-3 inhibtors and use there of, NAMPT OF COMPLEX I INHIBTORS OR USE THERE OF; Beigene: Honoraria; Merck: Honoraria; Janssen: Honoraria; AstraZeneca: Honoraria, Research Funding; Abbvie: Honoraria; CancerCare Manitoba Foundation: Research Funding; CIHR: Research Funding; LLSC: Research Funding; Lymphoma Canada: Research Funding; Hairy Cell Leukemia Foundation: Research Funding. Walewska:AbbVie, AstraZeneca, Janssen, Beigene: Other: meeting attendancies. Haferlach:MLL Munich Leukemia Laboratory: Current Employment, Other: Equity Ownership. Thieblemont:BMS/Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses, Research Funding; Janssen: Honoraria, Other: Travel Expenses; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses; Roche: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses, Research Funding; AbbVie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses; Gilead Sciences: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses; Hospira: Research Funding; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses; Cellectis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses; Kite: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; Incyte: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bayer: Honoraria; Paris University, Assistance Publique, hopitaux de Paris (APHP): Current Employment; Kyte, Gilead, Novartis, BMS, Abbvie, F. Hoffmann-La Roche Ltd, Amgen: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal